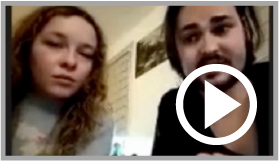
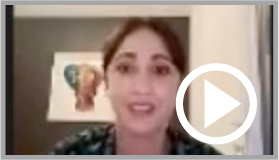
ELA international is giving a voice to the parents of sick children who are speaking out against the planned discontinuation of a treatment currently under trial
Scheduled discontinuation in 2024
In 2012, Takeda launched a clinical trial to evaluate the efficacy of an enzyme replacement therapy (TAK-611) in metachromatic leukodystrophy (MLD). In fall 2023, Takeda announced that the ongoing clinical trial had not meet its phase 2 endpoints, mainly based on the evaluation of patients’ motor functions. The major consequence of this declaration is the discontinuation of development and production of the TAK-611 molecule. Associations and investigators have been informed by the laboratory, and it is up to them to explain this to families.
ELA International gathers testimonies from parents of sick children, and from associations *
ELA International organized a meeting with the families concerned to collect their comments and opinions about the treatment on December 4, 2023. At the beginning of 2024, ELA International pursued to collect testimonials from various countries around the world: Argentina, Brazil, France, Germany, Japan, Poland, Spain and United States. Parents have been able to express their feelings and give their opinion on the effects they have noticed on their children’s quality of life. And everyone was unanimous. They all agree that their children suffer much less, are calmer, smile more, are more interactive and have more social activities, compared with untreated children.
ELA International contacted the President of Takeda
A document compiling all these testimonies has been sent by ELA International to the President of Takeda. It was accompanied by a letter calling for the observations made by families to be taken into consideration, and for the development of the treatment to be resumed with a clinical evaluation of the quality of life of MLD patients.
Takeda has listened to the patient voice
On March 18, 2024, Takeda responded to the entire MLD community, assuring that “Takeda has been actively engaged in discussions and listening to feedback from investigators, families and the patient advocacy community regarding the program.” Takeda representatives clearly heard that “families of the children in the trials have expressed a sincere desire to continue receiving TAK-611”.
As a result, Takeda has “taken the decision to extend the ongoing TAK-611 clinical trials until further notice. This extension of the clinical studies will include the continued supply of TAK-611 to existing trial sites. It will also allow patients and investigator sites to maintain their current follow-up”.
In addition, the company is working “to determine a more sustainable, long-term option in coordination with the relevant regulatory authorities.” ELA International would like to thank Takeda for its attentiveness and positive feedback and is committed to supporting any initiative taken in the interests of patients.
* With the support of the associations : ELA France, ELA Espagne ; ELA Allemagne ; ELA Italie ; ELA Suisse ; ELA Belgique ; ELA Luxembourg ; archangel ; Casa Hunter Institution ; Cure MLD ; Fundacion Lautaro te necesita ; MLD foundation ; MLD support UK ; ULF pour leur investissement.