Mathéo could have been cured with a gene therapy invented in France. He was denied treatment for commercial reasons.
To prevent this story from happening again, a mother of ELA calls out the French Ministers of Health and Research, as well as the Secretary General for Investment-France 2030 through a petition supported by the association.
Save children from being denied treatment for commercial reasons
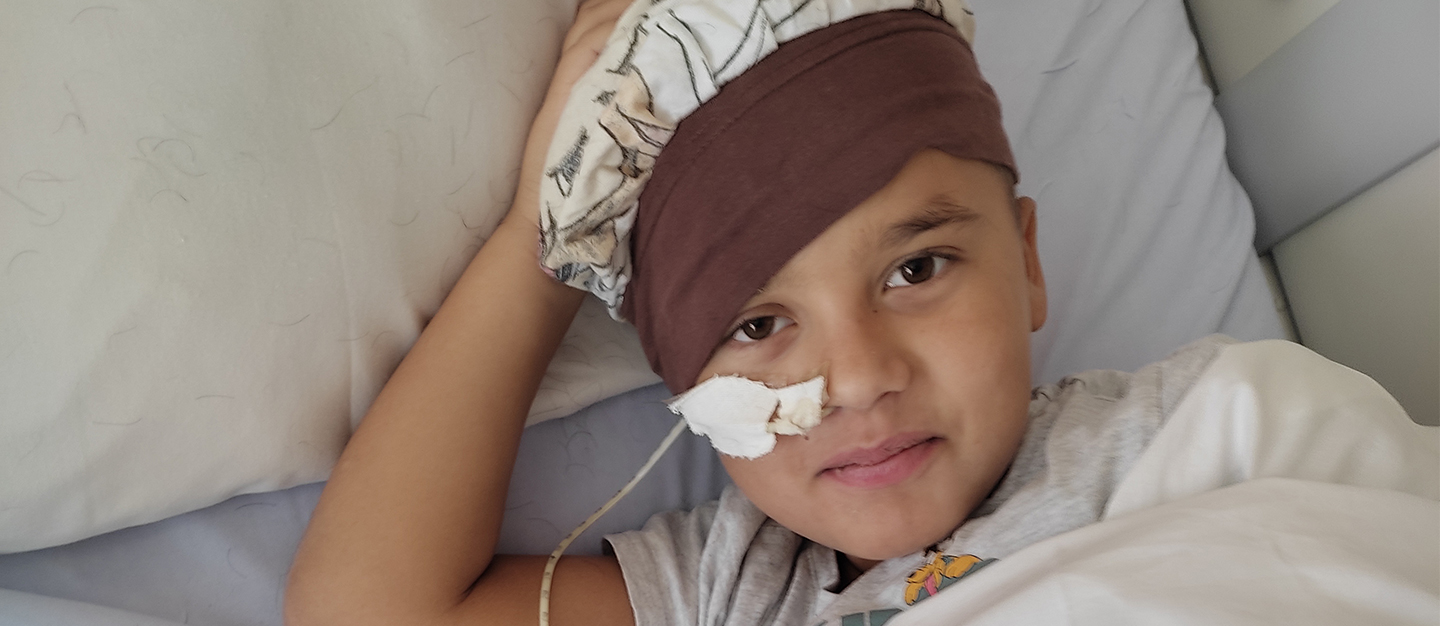
My name is Élodie Jourdan. I am the mother of a little Mathéo, now 8 years old. My son was in great shape until he started vomiting repeatedly. After further examination, we learned that he was a carrier of leukodystrophy, an extremely serious neurodegenerative disease that can destroy the brain of a previously healthy child within months. In November 2020, an MRI scan revealed a small lesion in his brain. Mathéo has no symptoms yet, but this sign indicates that the that the disease is progressing. It must be stopped at all costs. There is no time to lose.
So, the doctors offered us a brand new treatment, a gene therapy transplant called “Skysona”, which can cure Mathéo.
A transplant is a heavy treatment. But gene therapy has demonstrated its effectiveness and advantages over a conventional transplant: no need for a compatible donor, no risk of rejection, a rapid return to normality after 6 weeks in hospital, and no lifelong post-operative treatment.
We did not hesitate. We did all the necessary tests, and then, eight days before starting the transplant process, we got the bad news: the American laboratory that owns the marketing rights suddenly decided to leave France and Europe and to refocus on the American market for “commercial” reasons.
Mathéo will not benefit from this gene therapy…
Neither will the other European young ones… This is all the more unacceptable as this innovation was born in France with substantial funding from the European Leukodystrophy Association (ELA) and public research.
The blow is hard, between disappointment and inconsolable tears.
In the absence of Skysona, the doctors offered us a classic transplant to inject Mathéo with cells from a compatible donor. It took place in October 2021, but alas, it was clear that the transplant had been rejected. A nightmare. A second transplant was performed at the end of March 2022. But Mathéo was fragile, and infections attacked his heart, his kidneys, his spleen, his lungs and his liver. Our little boy fought, despite losing sight in one eye.
At the end of October 2022, we learned that the disease was continuing to progress despite the transplant, with new brain lesions appearing. We continue to hope that the transplant will eventually stop it for good. We’re keeping our fingers crossed, there’s nothing else we can do…
• How can we not be indignant when we know that this treatment is no longer available in Europe, even though Mathéo should have benefited from it?
• How can we not be indignant when we know that other gene therapies for other genetic diseases that are just as expensive are funded?
• How can we not anticipate the rapid and predictable increase in innovative treatments for rare diseases?
• How can we not be indignant that children are being condemned to general indifference when an effective treatment is available?
I don’t want our painful story to happen to other children and other parents. It is for them that I have decided to launch this petition. It is for them that I ask you to sign it en masse. Together we can still make a difference.
I have asked François Braun, Minister of Health, Sylvie Retailleau, Minister of Research, and Bruno Bonnell, Secretary General for Investment-France 2030:
• to create a special fund of up to 10 million euros in the first quarter of 2023, to enable children with leukodystrophy who could benefit from Skysona to be treated in the USA. This amount will have to be renewed as long as a permanent solution on French or European territory has not taken over.
• to produce Skysona in France, either in consultation with the American laboratory or through an ex-officio licence that would allow a third party to produce it.
Please support massively this mother’s initiative by signing the petition and share it on the social networks.